Study led by UM School of Medicine Faculty at the Center for Vaccine Development and Global Health Met safety goals and demonstrated efficacy
 Malaria remains one of the leading causes of death among children in sub-Saharan Africa, claiming more than 600,000 lives each year worldwide, with limited efficacy in currently available treatments and vaccines. Now a new early clinical trial found that a new monoclonal antibody demonstrated dose-dependent effect against the malaria parasite with minimal side effects.
Malaria remains one of the leading causes of death among children in sub-Saharan Africa, claiming more than 600,000 lives each year worldwide, with limited efficacy in currently available treatments and vaccines. Now a new early clinical trial found that a new monoclonal antibody demonstrated dose-dependent effect against the malaria parasite with minimal side effects.
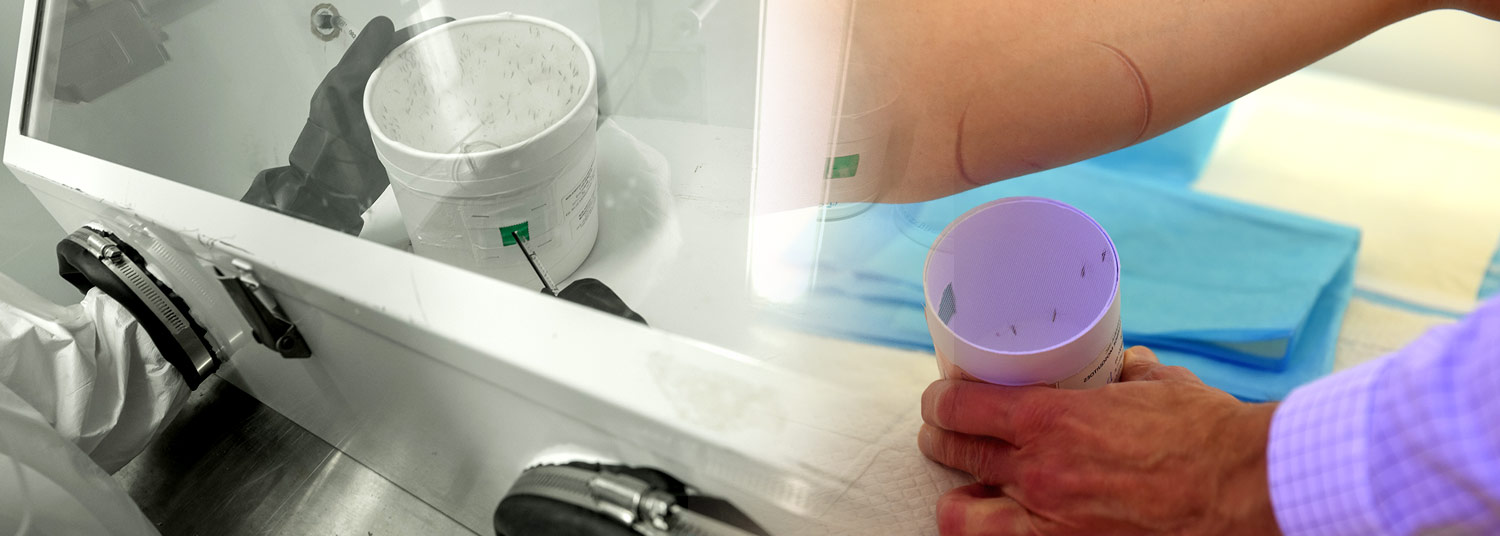
Researchers at the University of Maryland School of Medicine’s Center for Vaccine Development and Global Health (CVD) conducted the trial with healthy volunteers who were exposed in a controlled manner to bites from mosquitoes infected with the malaria parasite.
The results were recently published in The Lancet. The researchers conducted the experiment, sponsored by the Gates Medical Research Institute, with funding from the Gates Foundation..
 “Despite great progress, malaria continues to devastate families and communities across Africa,” said study lead author Kirsten E. Lyke, MD, Professor of medicine at the UM School of Medicine and principal researcher at CVD. “This new monoclonal antibody could transform how we prevent malaria in young children and pregnant women. Unlike vaccines that may require multiple doses or boosters, a single injection of a long-acting antibody can provide immediate, months-long protection. It’s a fundamentally different way to stop infection before it starts.”
“Despite great progress, malaria continues to devastate families and communities across Africa,” said study lead author Kirsten E. Lyke, MD, Professor of medicine at the UM School of Medicine and principal researcher at CVD. “This new monoclonal antibody could transform how we prevent malaria in young children and pregnant women. Unlike vaccines that may require multiple doses or boosters, a single injection of a long-acting antibody can provide immediate, months-long protection. It’s a fundamentally different way to stop infection before it starts.”
Monoclonal antibodies (mAbs) are laboratory-produced protein clones that mimic the body’s natural immune defenses. MAM01 targets a highly conserved region of Plasmodium falciparum circumsporozoite protein – a protein on the parasite’s outer surface – to block infection before it reaches the bloodstream.
 The phase 1, double-blind, placebo-controlled trial enrolled 38 healthy adults aged 18 to 50 years with no prior malaria exposure. Participants received one dose of MAM01 or placebo and were then exposed to malaria-carrying mosquitoes several months after dosing. This was done under carefully controlled conditions known as a challenge study. After the malaria challenge, none of the participants who received the highest dose of the monoclonal antibody developed infection, compared with all participants in the placebo group.
The phase 1, double-blind, placebo-controlled trial enrolled 38 healthy adults aged 18 to 50 years with no prior malaria exposure. Participants received one dose of MAM01 or placebo and were then exposed to malaria-carrying mosquitoes several months after dosing. This was done under carefully controlled conditions known as a challenge study. After the malaria challenge, none of the participants who received the highest dose of the monoclonal antibody developed infection, compared with all participants in the placebo group.
“These early results suggest that this monoclonal antibody has the potential to provide reliable protection against malaria, which continues to disproportionately affect children living in low- and middle-income countries,” said the study’s co-author. Matthew B. Laurens, MD, MPH, Professor of Paediatrics and Director of the Malaria International Clinical Trials Unit at CVD. “This is an important proof-of-concept for the field and a step forward for health equity.”
 No treatment-related serious side effects occurred.
No treatment-related serious side effects occurred.
“Testing of this preventive treatment has already begun in young children in Uganda, based on the promising results of the first trial conducted here,” said the UM School of Medicine Dean Mark T. Gladwin, MD, who is also vice president for medical affairs at the University of Maryland, Baltimore (UMB), and the John Z. and Akiko K. Bowers Distinguished Professor.
Added James Campbell, MD, MS, Interim Director of the Center for Vaccine Development and Global Health: “This study represents real hope for millions of children at risk. CVD has been a global leader in malaria research for more than 50 years, and these findings advance our mission to eliminate this disease through innovative science.”
About the Center for Vaccine Development and Global Health
The University of Maryland’s Center for Vaccine Development and Global Health (CVD) is internationally recognized for its pioneering work on malaria, cholera, typhoid and other infectious diseases. For more than five decades, CVD researchers have partnered with communities around the world to develop, test and deliver life-saving vaccines that improve global health outcomes.
About the University of Maryland School of Medicine
The University of Maryland School of Medicine, established in 1807 as the first public medical school in the United States, continues today as one of the fastest-growing, top-tier biomedical research institutions in the world. The school has nearly $500 million in total research funding, 46 departments, centers and institutes, more than 2,200 students, and over 3,000 faculty members, including notable members of the National Academy of Medicine. As the largest public medical school in the DC/MD/VA region, faculty physicians work to help patients manage chronic diseases such as obesity, cancer, heart disease and addiction, while also working on cutting-edge research to address the most critical generational health challenges. In 2024, the school ranked #12 among public medical schools and #27 among all medical schools for R&D spending by the National Science Foundation. With a total operating budget of $1.3 billion, the school partners with the University of Maryland Medical Center to serve nearly 2 million patients annually. The school’s global reach extends worldwide, with research and treatment facilities in 33 countries. At Maryland, the School of Medicine is spearheading new initiatives in artificial intelligence and health computing, collaborating with the University of Maryland BioPark to develop new medical technologies and bioengineering projects. For more information, visit medschool.umaryland.edu.